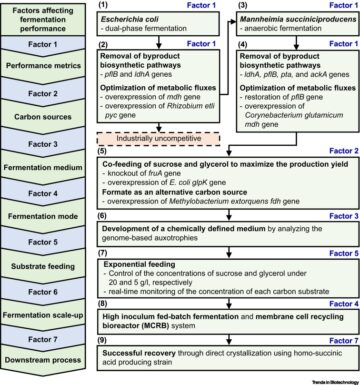
AAV gene therapy vectors and the challenges posed by neutralizing antibodies
]. Gene therapy as an approach treats or prevents a disease by modifying the underlying genetics. This can involve altering, repairing, or replacing a faulty gene to provide a functioning phenotype. Gene therapy has shown promise in preventing or treating certain inherited monogenic disorders (see Glossary) such as hemophilia, sickle cell disease, and Leber congenital amaurosis [
].
,
]. AAVs are small, non-enveloped viruses that are not replication competent unless in the presence of a helper adeno- or herpes virus []. Wild-type AAVs are not associated with any known human disease [
]. It is this lack of pathogenicity, combined with the ability to deliver up to approximately 4.8 kb of single-stranded (ss)DNA into a range of different tissue types that makes them an attractive vector for gene therapy delivery [
,
].
]. Clinically, no acute responses are typically observed.

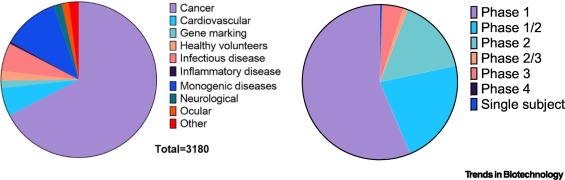
Figure 1Gene therapy trials by indication and phase (2021).

Figure 2Share of viral gene therapy trials by vector type (2021).
]. The immune response to AAV therapies is a complex picture and outside the scope of this review. For detailed reviews, please see [
,
,
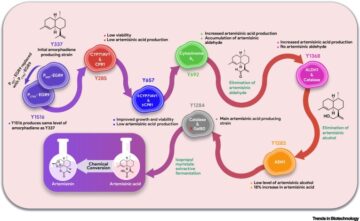
]. In short, once internalized, the virus is trafficked via endocytosis to the nucleus, where capsid proteins seek entry. The virus is then uncoated and the ssDNA is converted to double-stranded (ds)DNA, and in the case of therapeutic gene therapy, the relevant transgene is then expressed. Following transduction, AAV capsid proteins are broken down in proteasomes, which allows processing of peptides to MHC class I and MHC class II antigen-presenting molecules initiating T cell responses that lead to B cell activation and the production of anti-AAV IgG/neutralizing antibodies (NAbs) (Figure 3); the immune system is then primed to fight reinfection with AAV, essentially acting as a vaccine against re-administration of the therapy if required and reducing the therapeutic transduction efficiency.

Figure 3Illustration of adeno-associated virus (AAV) transduction and the humoral immune response.
,
,
]. Created with BioRender.com.
]. While undoubtedly a success, highlighting AAVs as an attractive choice for gene therapy vectors, the presence of high-titer NAbs remains a concern as it hinders the efficacy of future dosing and drastically reduces the number of patients the therapy can treat effectively.
], exposure to AAV occurs naturally and often early in life [
]. The seroprevalence of anti-AAV NAbs varies for differing serotypes and geographically between 15% and 60% [
,
,
]. Equally, although AAV vectors demonstrated long-term persistence in a clinical setting [
,
], due to the non-integrative mode of action, it is possible that a further dose is required later in a patient’s life to ensure therapeutic levels of transgene expression. Finding a safe and effective method or technology to overcome and evade AAV neutralization by the immune system will allow efficient and successful curative gene therapy delivery with lifelong therapeutic benefit to the greatest number of patients possible.
,
]. Currently, clinical trials exclude seropositive patients [
], vastly limiting the pool of patients the therapies are able to treat. The ideal successful technology would allow the treatment of seropositive patients while dampening the immune response to the vector and allowing future re-administration. At the same time, ideal immunosuppression methods, recently reviewed in [
] and summarized in Figure 4, require a favorable benefit/risk profile.

Figure 4Overview of approaches discussed in relation to the three main areas of focus to increase the treatable population.
Approaches discussed are grouped into the three desired attributes of allowing vector re-administration, enabling treatment of seropositive patients, and having a low immune burden. Exclusion of seropositive patients has a low immune risk but does not treat seropositive patients or allow vector re-administration. Novel and modified capsids are low immune risk and allow treatment of seropositive patients, but not vector re-administration. Adeno-associated virus (AAV) exosomes have a low immune burden and allow vector re-administration but could not treat seropositive patients. Broad-spectrum plasmapheresis, immune suppression, balloon catheter delivery, and swamping neutralizing antibodies (NAbs) with decoy or empty capsids allows treatment of seropositive patients but comes with an increased immunogenic risk to the patient. Of the techniques discussed, only specific immune suppression and plasmapheresis satisfy all three criteria.
,
,
], reinforcing the urgency to reduce the interactions between the AAV vector and the immune system and potentially rethink the single-large-dose approach to treatment [
].
In this review we focus on current and emerging technologies in the field to overcome and evade the immune response enabling AAV-mediated gene therapy to fulfil its potential, bringing lifelong curative treatment to patients with monogenic disorders.
Evading immune response and treating seropositive patients
]. It is clear that this is not applicable for other routes of administration to target tissues, particularly when AAV vectors are dosed systemically. An approach focusing on avoiding AAV contact with NAbs, however, does have benefits. A successful example of this evasion approach was performed through injection of AAV8 into the portal vein branch via balloon catheter that allowed hepatocyte transduction in the presence of low-titer NAbs. Although not applicable in the presence of higher-titer NAbs, and so far not applied in tissues other than liver, it is encouraging that local NAb avoidance can be suitable in some cases of tissue targeting.
]. This approach greatly increases the dose required and the associated manufacturing costs as well as the immunogenic burden.
]. It is clear that, where tissue tropism allows, vector serotype selection is important. However, even for the least immunogenic serotypes, there are many documented cases of pre-existing immune response [
,
,
,
,
,
,
].
,
,
]. This approach had varied effectiveness depending on the location of the capsid mutations. It is also possible that these mutations may alter tissue tropism and ultimately reduce transduction [
]. This does not overcome the challenge of vector re-administration unless used in combination with immune suppression [
], as cross-reactivity of NAbs across serotypes can still be observed and serotypes must be engineered to ensure they avoid all NAbs [
].
].
]. A challenge of these modifications is controlling the location of the functionalization reaction. This can be overcome using genetic code expansion tools, allowing the introduction of non-naturally occurring amino acids in the capsid at specific locations [
], which has been utilized to enable site-specific PEGylation showing reduced antibody recognition in vitro and reduced immune response in a rat model [
]. While PEGylation reduces the humoral response, AAV is still subject to cell-mediated immunity; to overcome this, there is the potential for the conjugation of immunosuppressive materials to AAV vectors. As demonstrated with immunosuppressive zwitterionic phosphoserine (PS)-containing polypeptide conjugated to AAV8, maintaining transduction efficiency and original tissue tropism and suppression of anti-AAV production in a mouse model [
] opened an avenue for targeted localized immune suppression, removing the increased infection risk of traditional systemic suppression.
]. This was performed by pelleting AAV-associated microvesicles by differential centrifugation of the production bioreactor supernatant. Although the mechanism by which microvesicle-associated AAV travels from the cell surface to the nucleus is not known, they were able to successfully transduce cells in the presence of a robust concentration of NAbs (1:2000), much higher than is typically seen in patients. Although higher NAb titers were still able to block the transduction, the resistance to neutralization shown by vesicle-associated AAVs is promising [
].
Preventing NAb generation to allow re-administration of vector
]. Although promising, the full picture of how long transduction may last is unclear and may vary depending on the target tissue and the age of the patient treated. Also, in some diseases capable of drastic reduction of life expectancy when untreated, a treatment early in life may be required [
]. In these instances, re-administration of vector may be needed in the future.
]. A benefit of this is that it uses already-approved therapies and drugs that can be selected based on the immune system components targeted (Table 1) . A combination of ciclosporin A, calcineurin inhibitor lowering T cell activity, and rituximab targeting B cells demonstrated a reduction in NAbs allowing vector re-administration in non-human primate (NHP) model [
] and this has been shown to be partially effective in reducing B cell responses in humans [
]. As rituximab is unable to target plasma cells, which do not express the CD20 antigen directly, it is not applicable to those with pre-existing immunity [
].
]
]. Further, when formulated in nanoparticles, rapamycin has shown greater efficacy in preclinical models in reducing NAbs and allowing vector re-administration in the primate model [
]. These approaches appear promising, particularly for re-administration to previously seronegative patients. As with rituximab and other pharmacological interventions are ineffective at bringing those previously exposed to wt-AAV into the treatable patient pool. The animal models studied must be seronegative to begin with.
Unfortunately, all forms of immune suppression come with the increased risk of infection. Combined with the therapies designed to treat potentially already chronically ill patients, great care must be applied when judging the risk–benefit analysis of immune suppression versus the practicality of gene therapy treatment.
]. CRISPR-Cas9 technology enables a platform to temporarily downregulate the MyD88 gene, briefly dampening the immune response, allowing administration of the vector and subsequently reducing the level of NAbs generated in response when observed in the mouse model [
]. As this reduces the immune response, it does not allow the treatment of seropositive patients. However, it may allow re-administration of vector in future and could be used in conjunction with novel serotype vectors or those with a lower prevalence of NAbs.
Temporary reduction of NAbs
]. In a study of ten patients seropositive for AAV1, 2, 6, and 8, up to five cycles of plasmapheresis reduced NAb titers in some patients to undetectable (<1:2) or low (≤1:5 to 1:2) levels [
], which would allow treatment of seropositive patients and potential delivery of the vector again if required in the future. Two cycles of plasmapheresis were sufficient to reduce NAb levels in a NHP model, enabling successful transduction comparable with seronegative animals [
], supporting the hypothesis that plasmapheresis may permit successful treatment in patients with pre-existing immunity.
This approach, however, requires a risk–benefit analysis similar to that discussed for immunosuppression, with the risk of depletion of all IgGs nonspecifically, the burden associated with multiple cycles of plasmapheresis ultimately leaving the patient more vulnerable to infections.
,
]. A significant challenge to the development of these tools is that NAbs are a polyclonal population, which varies serotype to serotype and between different patient populations. The binding efficiencies of some NAbs may differ in free AAV versus immobilized AAV. This approach is, however, highly promising, but requires consideration of ligand coupling density versus capacity, resin reuse cycles, and ligand leakage to ensure feasibility and scaling into larger animal models and then humans.

Figure 5Diagram highlighting the removal of neutralizing antibodies (NAbs) by specific plasmapheresis while retaining other IgGs.
Illustration of specific plasmapheresis, showing plasma containing a seropositive IgG pool passing through a pre-plasmapheresis column and the seronegative plasma post-column that can be returned to the patient.
]. The method sufficiently reduces the NAb titer to allow successful transduction of liver tissue in both mice and NHPs and crucially enabled subsequent successful re-administration of the vector [
,
]. In vitro testing on human plasma has also shown digestion of IgG, reducing anti-AAV8 titers even in the presence of anti-IdeS antibodies that are diffused in the human population [
]. As with nonspecific plasmapheresis, this approach depletes all IgG indiscriminately increasing the risk of infection; however, the safety is already demonstrated in patients with transplants. Therefore, although not without risks, IdeS provides a less invasive approach than plasmapheresis.
Glossary
Adeno-associated virus (AAV)
a small, non-enveloped virus able to deliver approximately 4.8 kb of linear ssDNA.
Immunosuppression
a treatment that reduces the efficacy of the immune system. In the case of gene therapy, immunosuppression has been shown to improve vector efficacy.
Monogenic disorder
attributable to a variation in a single gene, thus being a suitable target for treatment by gene therapy.
Neutralizing antibodies (NAbs)
antibodies generated in response to an antigen by the humoral response of the adaptive immune system. Anti-AAV NAbs prevent the virus from transducing cells.
Plasmapheresis
an extracorporeal technique involving the removal of blood plasma from a patient, the exchange of some components, in this case IgGs, and its return to the patient.
Serotype
a distinctly different variant of a bacterial or viral species. There are 13 naturally occurring wild-type AAV serotypes varying in capsid morphology and differing in tissue tropism.
Transduction
the process by which a vector transfers genetic material to a cell; in the case of gene therapy, this will be the therapeutic transgene.
- SEO Powered Content & PR Distribution. Get Amplified Today.
- Platoblockchain. Web3 Metaverse Intelligence. Knowledge Amplified. Access Here.
- Source: https://www.cell.com/trends/biotechnology/fulltext/S0167-7799(22)00303-1?rss=yes
- 1
- 10
- 100
- 11
- 15%
- 2020
- 2021
- 28
- 7
- 84
- 9
- a
- ability
- Able
- Accounts
- Achieve
- achieved
- across
- Action
- Activation
- activity
- Additional
- administered
- administration
- adopt
- Adult
- adults
- advance
- advanced
- ADvantage
- After
- against
- agency
- All
- Alliance
- Allowing
- allows
- alone
- already
- alternative
- Although
- analysis
- and
- animal
- animals
- Annex
- appear
- applicable
- applications
- applied
- approach
- approaches
- approved
- approximately
- areas
- Associate
- associated
- attractive
- attributes
- authors
- available
- Avenue
- avoiding
- based
- being
- benefit
- benefits
- between
- Billion
- Block
- blocking
- blood
- Branch
- Breakdown
- briefly
- Bringing
- Broken
- burden
- Cancer
- candidates
- capable
- Capacity
- care
- case
- cases
- causes
- Cells
- centered
- certain
- challenge
- challenges
- characteristics
- Chart
- chemical
- Children
- choice
- circulating
- class
- clear
- Clinical
- clinical trials
- Close
- code
- Cohort
- Column
- combination
- combined
- come
- Common
- commonly
- comparable
- compared
- competent
- complex
- components
- concentration
- Concern
- Concerns
- consideration
- considering
- contact
- controlling
- converted
- Costs
- could
- countries
- coupled
- created
- CRISPR
- criteria
- critical
- crucially
- Current
- Currently
- cycles
- data
- Database
- deaths
- deliver
- delivery
- demonstrated
- demonstrating
- Depending
- Derived
- designed
- detailed
- Development
- differ
- different
- differing
- directly
- discussed
- Disease
- diseases
- disorders
- distinct
- divided
- dna
- down
- drastically
- drug
- Drugs
- each
- Early
- Effective
- effectively
- effectiveness
- efficiencies
- efficiency
- efficient
- eliminates
- emerged
- emerging
- enable
- enables
- enabling
- encouraging
- enhanced
- ensure
- entry
- equally
- essentially
- Ether (ETH)
- Even
- evolution
- evolved
- example
- exchange
- expansion
- exposed
- Exposure
- express
- expressed
- eye
- factors
- faulty
- field
- fight
- Figure
- finding
- First
- Focus
- focusing
- followed
- following
- forms
- Forward
- Free
- from
- full
- functioning
- further
- future
- generate
- generated
- generation
- Genetics
- given
- great
- greater
- greatest
- greatly
- Growth
- having
- healthy
- High
- higher
- Highlighted
- highlighting
- highly
- hinders
- hold
- How
- However
- HTTPS
- human
- Humans
- ideal
- identified
- Immune system
- immunity
- Impact
- implications
- important
- imposed
- improve
- in
- Increase
- increased
- Increases
- increasing
- influenced
- info
- information
- initial
- innate
- interact
- interactions
- interior
- Introduction
- investment
- involve
- IT
- journal
- kidney
- known
- Label
- Lack
- larger
- Last
- lead
- leaving
- Level
- levels
- Life
- limitation
- List
- little
- Liver
- local
- location
- locations
- Long
- long-term
- Low
- Main
- major
- MAKES
- manufacturing
- many
- material
- materials
- Meanwhile
- mechanism
- medicine
- method
- methods
- Mode
- model
- models
- Modifications
- modified
- more
- most
- mRNA
- multiple
- NAB
- Need
- negative
- New
- novel
- number
- ONE
- ongoing
- opened
- Opportunity
- Option
- original
- Other
- outside
- Overcome
- particularly
- Passing
- passive
- patient
- patients
- Peg
- perform
- perhaps
- persistence
- phase
- Physically
- picture
- Plasma
- platform
- plato
- Plato Data Intelligence
- PlatoData
- playing
- please
- pool
- population
- populations
- Portal
- possible
- potential
- potentially
- presence
- presented
- prevalent
- prevent
- preventing
- Prevention
- previous
- previously
- Prior
- process
- processing
- Produced
- Product
- Product information
- Production
- Products
- Profile
- Profiles
- promise
- promising
- Protein
- Proteins
- provide
- provided
- provides
- range
- rapamycin
- RAT
- reaction
- ready
- recent
- recently
- recognition
- recognizes
- reduce
- Reduced
- reduces
- reducing
- regenerative
- registered
- regular
- regulator
- relation
- relevance
- relevant
- remains
- removal
- remove
- removing
- repairing
- repeat
- replication
- report
- Reported
- require
- required
- requires
- resilience
- Resistance
- Respond
- response
- retaining
- Retina
- return
- review
- reviewed
- Reviews
- Risk
- risks
- robust
- Role
- routes
- safe
- Safety
- same
- scaling
- scope
- screening
- Seek
- selected
- selection
- selective
- Sequence
- Serum
- setting
- Short
- shown
- significant
- similar
- single
- small
- So
- so Far
- some
- specific
- specificity
- Stability
- State
- Still
- strategies
- Strategy
- structure
- studied
- studies
- Study
- subject
- subsequent
- Subsequently
- success
- successful
- Successfully
- such
- sufficient
- suitable
- SUMMARY
- Supporting
- suppression
- Surface
- Symptoms
- system
- systemic
- T cells
- Target
- targeted
- targeting
- techniques
- Technologies
- Technology
- temporary
- ten
- Terminal
- Testing
- The
- therefore
- three
- Through
- time
- to
- tools
- top
- traditional
- transfer
- transfers
- Translation
- travels
- treat
- treating
- treatment
- trial
- trials
- true
- types
- typically
- Uk
- Ultimately
- underlying
- understanding
- undoubtedly
- urgency
- use
- utilized
- Vaccine
- Variant
- Versus
- via
- virus
- viruses
- Vulnerable
- W
- ways
- which
- while
- will
- Wilson
- without
- worldwide
- would
- X
- years
- yields
- zephyrnet