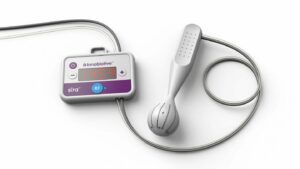
Cognito Therapeutics has enrolled the first patient in a biomarker substudy within its HOPE pivotal trial evaluating sensory stimulation device in Alzheimer’s disease patients.
The HOPE study (NCT05637801) is assessing the efficacy of Cognitio’s GammaSense Stimulation system in subjects with mild to moderate Alzheimer’s disease. The randomised, double-blind, sham-controlled, adaptive-design trial aims to administer the therapy to 530 subjects with Alzheimer’s for 12 months.
The substudy will evaluate magnetic resonance imaging (MRI), cerebrospinal fluid and plasma biomarkers. The main study will measure the efficacy of Alzheimer’s disease tests such as Alzheimer’s Disease Cooperative Study-Activities of Daily Living (ADCS-ADL).
Cognito’s therapy is unlike much of the Alzheimer’s development in the pharma industry. The non-pharmacological approach involves eliciting gamma-frequency brain activity through non-invasive visual and audio-sensory stimulation via a headset, which the patient can use at home. The US-based company says its technology has the potential to slow the progression of Alzheimer’s disease.
A market model by GlobalData estimates the global neuromodulation device market will be worth $11.4bn by 2033, up from $6bn in 2022.
Cognito uses the idea that gamma-frequency brain activity – essential for brain function – is impaired in those with Alzheimer’s. The company says that evaluating biomarkers in the substudy will help with earlier predictive responses to Cognito therapy and better identify patients who respond best to treatment.
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Your download email will arrive shortly
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataCognito’s device, which was granted breakthrough device designation by the US Food and Drug Administration (FDA) in January 2021, made waves when data demonstrated it reduced brain atrophy and ADCS-ADL decline, with no amyloid-related imaging abnormalities (ARIA) or treatment-limited serious adverse events. The data was from Cognito’s Phase II OVERTURE study (NCT03556280), with positive results announced by the company in October 2023.
In March 2023, Cognito secured $73m in a Series B financing round, with backing from the Alzheimer’s Drug Discovery Foundation, to advance clinical development of the device.
Cognito’s chief medical officer Ralph Kern said: “We are on track to achieve study completion within our projected timelines, with over 240 patients currently enrolled in the HOPE study across 60 active sites across the US.
“We are well-positioned to achieve our study timeline goals, as we advance towards potential approval of the first non-pharmacologic disease-modifying therapy for Alzheimer’s disease.”
Elsewhere in the medtech space, Electro Cellular Healthcare Solutions (ECHS) is also trialling a wearable headband device to treat patients with mild to moderate Alzheimer’s disease.
<!-- -->- SEO Powered Content & PR Distribution. Get Amplified Today.
- PlatoData.Network Vertical Generative Ai. Empower Yourself. Access Here.
- PlatoAiStream. Web3 Intelligence. Knowledge Amplified. Access Here.
- PlatoESG. Carbon, CleanTech, Energy, Environment, Solar, Waste Management. Access Here.
- PlatoHealth. Biotech and Clinical Trials Intelligence. Access Here.
- Source: https://www.medicaldevice-network.com/news/cognito-starts-biomarker-substudy-in-alzheimers-device-trial/
- :has
- :is
- $UP
- 11
- 12
- 12 months
- 2021
- 2022
- 2023
- 60
- 7
- 8
- 9
- a
- About
- Achieve
- across
- active
- activity
- administer
- administration
- advance
- adverse
- aims
- Alzheimer’s
- an
- and
- announced
- approach
- approval
- ARE
- aria
- article
- AS
- Assessing
- At
- b
- backing
- banner
- BE
- below
- beneficial
- BEST
- Better
- biomarker
- biomarkers
- Brain
- Brain activity
- breakthrough
- business
- by
- CAN
- cellular
- chief
- Clinical
- company
- competitive
- completion
- comprehensive
- confident
- cooperative
- credit
- Currently
- daily
- Daily news
- data
- decision
- Decline
- demonstrated
- designation
- Development
- device
- discovery
- Disease
- download
- drug
- drug discovery
- Earlier
- Edge
- efficacy
- end
- enrolled
- essential
- estimates
- evaluate
- evaluating
- events
- fda
- financing
- First
- fluid
- food
- Food and Drug Administration
- Food and Drug Administration (FDA)
- For
- form
- Foundation
- Free
- from
- function
- Gain
- Global
- GlobalData
- Goals
- granted
- Headset
- healthcare
- help
- Home
- hope
- HOURS
- However
- HTTPS
- ICON
- idea
- identify
- ii
- image
- Imaging
- in
- industry
- Industry Insights
- insights
- involves
- IT
- ITS
- January
- January 2021
- jpg
- leading
- living
- made
- Main
- make
- March
- Market
- measure
- medical
- Medtech
- mild
- model
- moderate
- months
- most
- MRI
- much
- news
- Newsletter
- no
- october
- of
- offer
- Officer
- on
- or
- our
- Our Company
- over
- patient
- patients
- Pharma
- phase
- pivotal
- Plasma
- plato
- Plato Data Intelligence
- PlatoData
- potential
- powered
- predictive
- Profile
- Profiles
- progression
- projected
- quality
- Reduced
- research
- resonance
- Respond
- responses
- Results
- round
- Said
- Save
- says
- Secured
- Series
- Series B
- serious
- Sites
- slow
- So
- Solutions
- Space
- start
- starts
- Study
- such
- SVG
- system
- Technology
- tests
- that
- The
- therapeutics
- therapy
- this
- those
- Through
- timeline
- timelines
- to
- towards
- track
- treat
- treatment
- trial
- unique
- unlike
- us
- US Food
- use
- uses
- via
- visual
- want
- was
- waves
- we
- wearable
- when
- which
- WHO
- will
- with
- within
- worth
- you
- Your
- zephyrnet