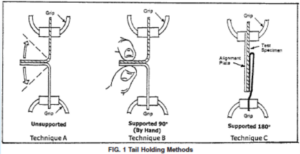
Rob Packard is hosting the Auditing QSR and MDSAP requirements four-part webinar series from August 9th to 30th, 2023.
Your cart is empty
Register for the 4-part Webinar Series on Auditing QSR & MDSAP requirements for $299
This is a 4-part webinar series that will be conducted live on August 9, 17, 23, and 30 (2023) via Zoom. The second webinar was rescheduled from 16th to 17th due to a scheduling conflict. This course assumes that the participant already has experience auditing to ISO 13485 and/or ISO 9001.
Price: $299.00
Outline for the Auditing QSR & MDSAP requirements webinar series
Registrants will receive a confirmation email because we deliver content and notification of updates through AWeber as an email subscription. After confirmation, you will receive login information for the four live Zoom webinars. Each of the four webinars will be approximately 45 minutes in duration, and the training content is organized as follows:
Auditing QSR & MDSAP requirements Kick-off – Wednesday, August 9, 2023, @ 11:00 am EDT
- Detailed Agenda TBD
Session 2 – Thursday, August 17, 2023, @ 11:00 am EDT (rescheduled to 17th due to conflict)
- Detailed Agenda TBD
Session 3 – Thursday, August 23, 2023, @ 11:00 am EDT
- Detailed Agenda TBD
Auditing QSR & MDSAP requirements Finale – Wednesday, August 30, 2023, @ 11:00 am EDT
- Detailed Agenda TBD
About Your Medical Device Academy Webinar Instructor Rob Packard

Rob Packard is a regulatory consultant with ~25 years of experience in the medical device, pharmaceutical, and biotechnology industries. He is a graduate of UConn in Chemical Engineering. Rob was a senior manager at several medical device companies—including the President/CEO of a laparoscopic imaging company. His Quality Management System expertise covers all aspects of developing, training, implementing, and maintaining ISO 13485 and ISO 14971 certifications. 2009-2012, he was a lead auditor and instructor for one of the largest Notified Bodies. Rob’s specialty is regulatory submissions for high-risk medical devices, such as implants and drug/device combination products for CE marking applications, Canadian medical device applications, and 510(k) submissions. The most favorite part of his job is training others. He can be reached via phone at 802.281.4381 or by email. You can also follow him on YouTube, LinkedIn or Twitter.
Posted in: Uncategorized
- SEO Powered Content & PR Distribution. Get Amplified Today.
- PlatoData.Network Vertical Generative Ai. Empower Yourself. Access Here.
- PlatoAiStream. Web3 Intelligence. Knowledge Amplified. Access Here.
- PlatoESG. Automotive / EVs, Carbon, CleanTech, Energy, Environment, Solar, Waste Management. Access Here.
- BlockOffsets. Modernizing Environmental Offset Ownership. Access Here.
- Source: https://medicaldeviceacademy.com/auditing-qsr/
- :has
- :is
- 1
- 11
- 17
- 2023
- 23
- 30
- 30th
- 49
- 7
- 9
- 9th
- a
- Academy
- After
- agenda
- All
- already
- also
- am
- an
- and
- applications
- approximately
- AS
- aspects
- assumes
- At
- auditing
- AUGUST
- author
- BE
- because
- biotechnology
- bodies
- by
- CAN
- Canadian
- certifications
- chemical
- COM
- combination
- company
- conducted
- confirmation
- conflict
- consultant
- content
- course
- covers
- Date
- deliver
- developing
- device
- Devices
- due
- duration
- each
- end
- Engineering
- Ether (ETH)
- experience
- expertise
- Favorite
- finale
- follow
- follows
- For
- four
- from
- graduate
- he
- high-risk
- him
- his
- Home
- hosting
- http
- HTTPS
- Imaging
- implementing
- in
- industries
- information
- ISO
- ISO 9001
- Job
- jpg
- kick-off
- largest
- lead
- LINK
- live
- login
- maintaining
- management
- management system
- manager
- marking
- medical
- medical device
- medical devices
- Metadata
- minutes
- most
- notification
- of
- on
- ONE
- or
- Organized
- Others
- page
- part
- Pharmaceutical
- phone
- plato
- Plato Data Intelligence
- PlatoData
- posted
- Posts
- Preview
- Products
- Profile
- public
- quality
- reached
- receive
- regulatory
- Requirements
- rob
- scheduling
- Second
- senior
- Series
- several
- Specialty
- Submissions
- subscription
- such
- system
- that
- The
- this
- Through
- thursday
- to
- Training
- updated
- Updates
- usb
- via
- Video
- was
- we
- webinar
- Webinars
- Wednesday
- will
- with
- years
- you
- Your
- youtube
- zephyrnet
- zoom